
Dr BRIENNE MCKENZIE
STUDY OF TARGET CELL PYROPTOSIS IN THE CONTEXT OF
CTL-INDUCED DEATH
Cytotoxic T lymphocytes (CTLs) are believed to kill their targets through a programmed, non-inflammatory cell death mechanism known as apoptosis. However, new evidence has also highlighted the potential role of pyroptosis, a highly inflammatory cell death mechanism that is executed by pore-forming proteins called gasdermins. We hypothesize that, through the combined release of soluble cytotoxic molecules and supra-molecular attack particles (SMAPs), CTLs activate both apoptotic and pyroptotic signaling cascades in tumor cells in parallel, to bypass cell death resistance mechanisms. The objectives of this project are: (i) to study the activation of gasdermin family proteins in response to CTL and/or SMAP attack (ii) to identify molecular triggers upstream of pyroptosis, (iii) elucidate the cellular mechanisms of pyroptosis resistance in tumor cells and (iv) measure pyroptosis in tissues in the context of tumor-infiltration by CTLs and relate these observations to clinical outcomes. Understanding the molecular mechanisms by which cells die in response to CTL and SMAP attack will allow us to determine molecular resistance mechanisms that reduce the efficiency of CTL-mediated tumor killing, identify predictive biomarkers of resistance, and design rational combination therapies to improve tumor cell susceptibility to CTL- and SMAP-induced death.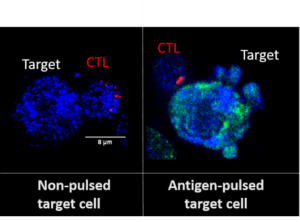
Figure 1 : Antigen-loaded target cells cleave and activate the pyroptosis executor protein, gasdermin D, after CTL attack.
Target cells are first loaded with antigen to induce CTL lethal hit delivery (right; no-antigen control shown on the left). Both CTLs (immunostained with perforin, red) and target cells constitutively express total gasdermin D (blue). However, after successful CTL attack, the activated form of the pyroptosis executor protein, gasdermin D, is found in dying target cells (green), which form pyroptotic bodies (bubbles) on the cell membrane during the final stages of cell death.
