Team
Etienne Chatelut
DIAD :
Dose Individualisation of Anticancer Drugs
The specificities
of our research axis
Pharmacokinetics
Pharmacogenetics
Individualisation of anticancer treatment
Therapeutic
Drug Monitoring
Liquid chromatography
Theranostic radiolabelled molecules
Family genetic predisposition
Research
projects
Oral therapies: Adaptation of oral anticancer drug doses in patients who cannot swallow.
Dr Cécile Arellano & Dr Florent Puisset
Pharmacokinetics and innovative therapeutics
Dr Ben Allal
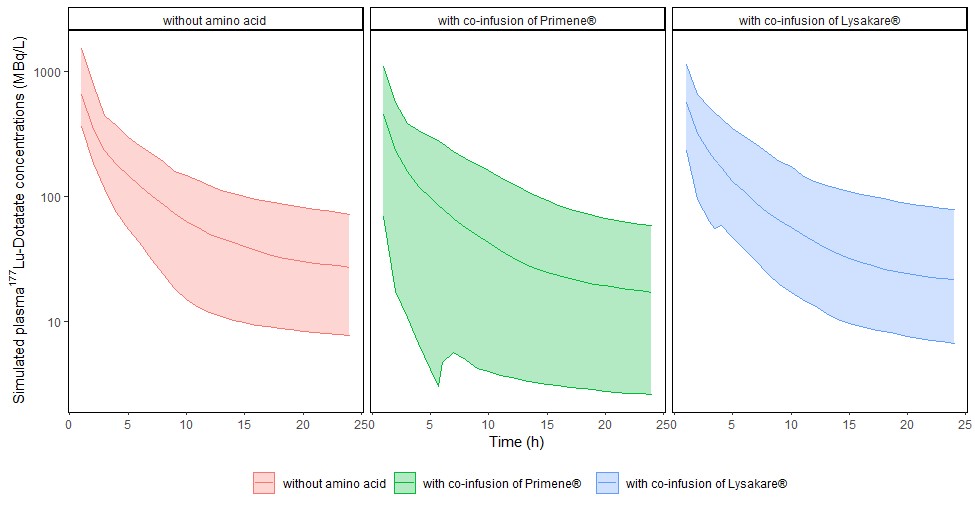
Pharmacokinetics and theranostic radiolabelled molecules.
Dr Lawrence Dierickx
Quantifying the sources of inter-individual variability in hormone therapy exposure
Dr Fabienne Thomas
Pharmacokinetic modelling of radiolabelled antibodies to understand resistance mechanisms in immunotherapy.
Dr Mélanie White-Koning
MATADOR and EXPECT projects: identification of new predictive factors for male breast cancer (MATADOR) and young colon cancer (EXPECT).
Dr Christine Toulas
THE TEAM’S
FOCUS
Discover
Understand
Participate
Scientific production
PUBLICATIONS 2026
PUBLICATIONS 2025
PUBLICATIONS 2024
PUBLICATIONS 2023
PUBLICATIONS 2022
PUBLICATIONS 2021
PUBLICATIONS 2020
PUBLICATIONS 2019
Team members
Partnerships & Funding

Toulouse Cancer Research Center (Oncopole)
Toulouse - FR
Follow us on social network
Contact us
+33 5 82 74 15 75
Want to join
the CRCT team ?