What is the role of autophagy in response to targeted therapy in ALK+ lymphomas? Does this process promote tumor cell survival or death?
Anaplastic large cell lymphoma
Oncogene ALK
Autophagy
Targeted therapy
Combined therapy
BCL-2
RAF-1
Cell survival
Cell death
Dr Sylvie Giuriato and Pr E. Espinos team T2i et RNAreg
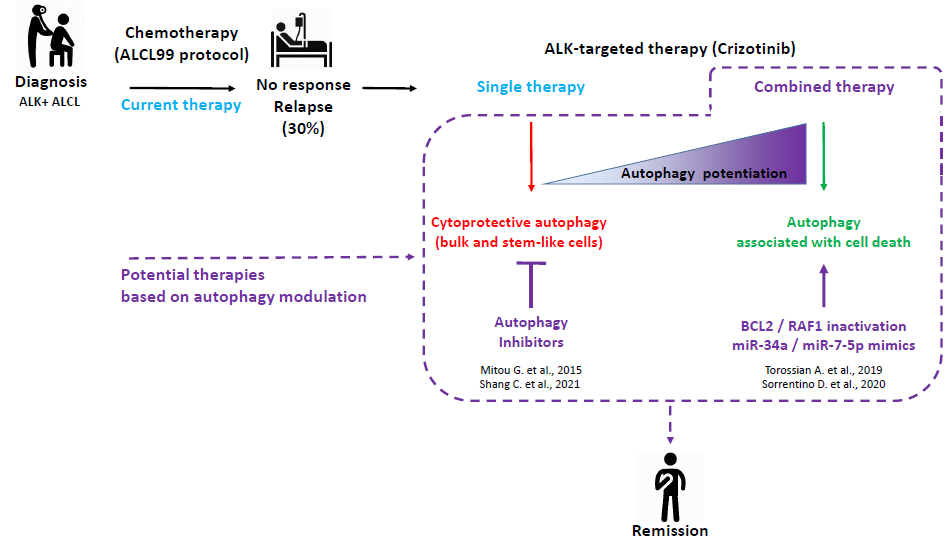
Current treatment of ALK+ lymphoma is not optimal as 30% of patients do not respond or relapse within 5 years of chemotherapy. In order to improve this situation, biomedical research has focused on the development of alternative therapies (targeted and combination therapies). This review summarizes our work on the therapeutic benefit of modulating autophagy (a physiological process of self-digestion of intracellular components) in the context of targeted anti-ABL therapy, using the molecule “Crizotinib”. Consistent with the known functional duality of autophagy (as a survival mechanism or as a mechanism associated with cell death), our previous work demonstrated that targeted treatment of ALK+ lymphoma cells with crizotinib alone induced protective autophagy, including in stem-like tumor cells, whereas treatments combining crizotinib with inhibition of other proteins (notably BCL-2 or RAF-1) potentiated autophagy and promoted cell death. The different molecular mechanisms known and described in the literature by which autophagy performs these protective or deleterious functions for tumor cells are also discussed.
Numerous works are in progress in the scientific and pharmaceutical communities for the development of specific autophagy modulating molecules (inhibitors or activators), for a use in clinical oncology.
In view of our results on the role of autophagy in the response of LAGC cells to crizotinib, it is conceivable that clinical trials combining a therapy targeting ALK with a molecule targeting autophagy (or one of its regulatory proteins) could be initiated.
This review details the current knowledge on the cytoprotective or cytotoxic role of autophagy in response to different therapies, data that could potentially be used to improve the treatment of ALK+ lymphomas. Several hypotheses on the molecular mechanisms underlying this functional duality are developed and are prospects for future research in the field of ALK+ lymphomas and autophagy.
Key collaborations and funding
Dr Raymond Lai: Department of Laboratory Medicine and Pathology, Department of Oncology, University of Alberta, Edmonton, Alberta, Canada; rlai@ualberta.ca
Funding: European Union Horizon 2020 Research and Innovation Programme under Marie Sklodowska-Curie Grant Agreement No. 675712.
Discover the published article
Review Cells. 2021 Sep 23;10(10):2517. doi: 10.3390/cells10102517.
The Dual Role of Autophagy in Crizotinib-Treated ALK + ALCL: From the Lymphoma Cells Drug Resistance to Their Demise
Estelle Espinos, Raymond Lai, Sylvie Giuriato
Collaborations and acknowledgements


Toulouse Cancer Research Center (Oncopole)
Toulouse – FR
Contact us
+33 5 82 74 15 75
Want to join
the CRCT team ?


