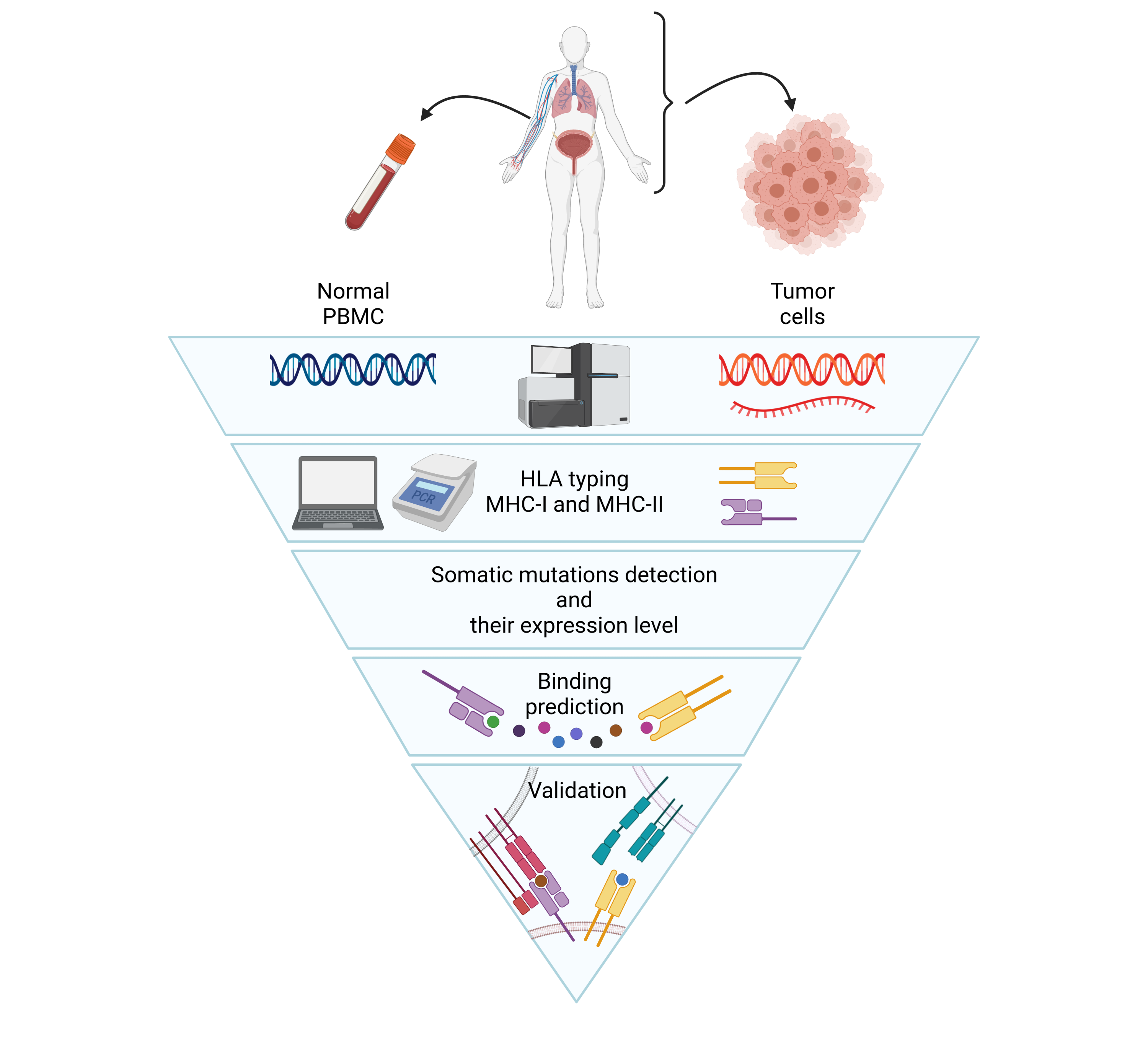
Role of tumour ag-specific T cells in the clinical response to immunotherapy.
Pr Maha Ayyoub
The success of anti-cancer immunotherapies (especially antibodies against immune checkpoints) relies on the recruitment and activation of cytotoxic T lymphocytes, specific to tumour antigens. These T cells exist in the bloodstream and at the tumour site as a continuum of activated and depleted lymphocytes, the latter being characterised by inhibition of the cytotoxic functions of these cells.
Autophagy is a highly conserved physiological process, allowing cells to maintain a cytoplasm free of toxic elements, as well as to meet their energy needs. While the role of autophagy in T cell development, activation and differentiation has been well described in the literature over the last decade, its role in T cell depletion is not yet clearly established.
Our projects aim to understand the role of autophagy in the T cell exhaustion program, as well as to evaluate whether targeting autophagy (inhibiting or activating the process) could improve the efficacy of immunotherapies targeting immune checkpoints, thus exhaustion. These projects will be addressed in three study models, in vitro, in vivo, as well as in human tumour biopsies, to understand :
- The impact of autophagy inhibition or activation on the acquisition of immune checkpoints, by performing cultures of T lymphocytes engaged in a chronic activation exhaustion programme.
- The status of autophagy in the immune microenvironment of tumours that progress or are rejected in orthotopic tumour models (already established in the laboratory for different cancers).
- The potential relationship between the autophagic activity of cytotoxic T lymphocytes and their ability to respond or not to immunotherapies. This will be investigated by bioinformatics analysis using single cell RNAseq data obtained on TILs (tumour infiltrating lymphocytes) from patient tumour biopsies.
Thus, these studies should shed new light on the therapeutic modulation of autophagy in the improvement of anti-cancer immunotherapies.
Technologies used
Flow cytometry (17 colour staining); HLA/peptide multimer analysis of antigen-specific T cells; RNASeq, whole exome sequencing; Bioinformatics analysis; Functional analyses of T cells.