
CHRISTIAN TOURIOL
Role of endoplasmic reticulum stress in the progression and resistance of acute myeloid leukemia
Acute leukemias are hematological tumors characterized by the expansion of blood cell precursors in the bone marrow. Among these pathologies, acute myeloid leukemia is the most frequent in adults. Standard treatment of patients is quite effective in the short term but the rate of complete remission obtained is unsatisfactory and the survival rate remains low, especially for subjects over 60 years of age, due to the appearance of cells resistant to treatment. It is therefore crucial to unveil the cellular mechanisms of resistance that irremediably appear during treatment in order to allow the development of new therapeutic agents likely to improve the prognosis of subjects with relapsing acute myeloid leukemia.
The endoplasmic reticulum is a very important cellular compartment in which transmembrane and secreted proteins are made, folded and assembled (Figure 1). During tumor progression, the rapid and uncontrolled growth of cancer cells will lead to an alteration of the endoplasmic reticulum functions. This will induce a stress, called endoplasmic reticulum stress, which activates the triggering of a physiological response, called UPR (“Unfolded Protein Response”), allowing the adaptation of tumor cells to their environment and their survival. This adaptation program is relayed by three proteins located in the membrane of the endoplasmic reticulum: i) the IRE1 protein (“Inositol-Requiring Enzyme-1”) ii) the transcription factor ATF6 iii) the PERK protein (Protein Kinase R (PKR)-like ER Kinase)
This response has been extensively studied in solid tumors. However, the understanding of the role of UPR in the development and progression of acute myeloid leukemias is extremely patchy. The aim of our project is therefore to study, in a global way, but also by distinguishing each of the pathways (PERK, IRE1 and ATF6), the role of the UPR response on the tumor progression of acute myeloid leukemias and their resistance to treatments.
This work is performed in cellulo for the mechanistic aspects and in vivo in animal models. For each of the UPR pathways, we have established innovative cellular models allowing “loss” or more especially “gain of function” approaches. We hope to identify new potential therapeutic targets that could increase the efficacy of available treatments against acute myeloid leukemia and possibly against other tumor types in which reticulum stress is activated.
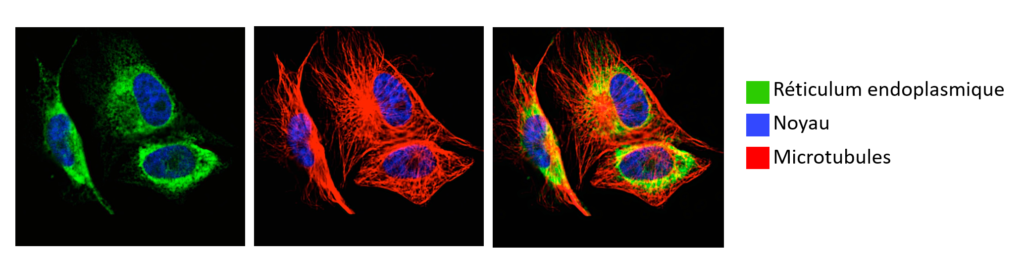
Legend: Fluorescence microscopy
Specific labeling of the endoplasmic reticulum (green), nucleus (blue) and microtubules (red) of a cancer cell
